Artigo 4: Versão Pro otimizada
Escolhendo pacotes de gel PCM para biotecnologia sem simplificar demais o pacote
Escolher bem nesta categoria se resume a um princípio: combine o pacote com a aplicação antes de comparar fornecedores. Muitas decisões de fornecimento começam com o tamanho da embalagem ou preço e só mais tarde descobrem problemas com o condicionamento, ajustar, vazamento, hipotermia, documentação, ou continuidade de fornecimento. Uma sequência melhor é definir primeiro o trabalho e depois auditar qual fornecedor pode repeti-lo de forma confiável.
Um PCM de 5°C é comumente escolhido quando o objetivo real é manter o produto dentro de uma faixa de 2-8°C sem expô-lo ao choque frio que pode ocorrer quando uma embalagem à base de água totalmente congelada fica diretamente contra a carga útil.
O que o produto deve fazer – e o que não se deve esperar que ele faça
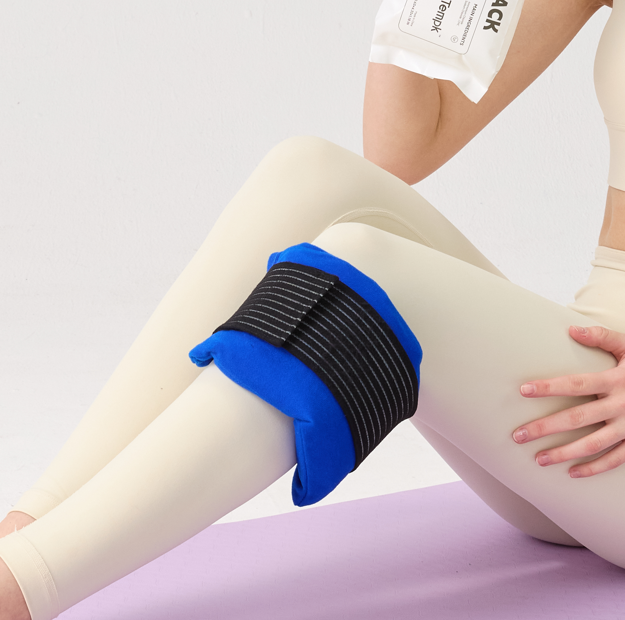
Um pacote de gel PCM para trabalho biotecnológico é um tampão térmico projetado para mudar de fase próximo a uma temperatura escolhida, em vez de se comportar como gelo comum.. Isso é importante quando a carga útil pode ser danificada por temperaturas muito baixas ou muito altas. Em termos práticos, o pacote é selecionado para suportar o reagente, amostra, conjunto, ou remessas biológicas que precisam de um controle mais rígido do que uma bolsa de gel congelada padrão sempre pode fornecer.
Os compradores atacadistas geralmente estão resolvendo um problema de gerenciamento de risco, não simplesmente preenchendo o espaço vazio da caixa. Eles precisam de um refrigerante que condicione de maneira previsível, cabe no contêiner isolado sem esmagar a carga útil, e oferece suporte a uma rotina de embalagem repetível em bancadas de embalagem movimentadas. É improvável que um fornecedor que não possa discutir esses detalhes operacionais seja forte o suficiente para a distribuição de biotecnologia.
Para pistas de biotecnologia, orientação pública sobre o PIB farmacêutico, expedidores passivos qualificados, e embalagens de substâncias biológicas deixam um ponto muito claro: a proteção de temperatura deve ser avaliada como um sistema de transporte completo que inclui isolamento, configuração de carga útil, duração do transporte, e monitoramento. A OMS e as orientações relacionadas com a cadeia de frio enfatizam a qualificação do sistema de transporte marítimo totalmente passivo ou ativo, incluindo condicionamento de refrigerante, massa de carga útil, perfil de rota, e exposição ambiental. No transporte de biotecnologia, esse limite é importante porque uma carga útil de alto valor pode falhar mesmo quando a especificação do refrigerante parece correta isoladamente.
Como o formato certo é escolhido
Os pacotes de gel padrão geralmente dependem de um gel à base de água ou de polímero que é congelado antes do uso. Eles são versáteis, econômico, e amplamente disponível em soft bags ou formatos mais estruturados. Pacotes PCM são mais especializados. Eles são projetados para absorver e liberar calor próximo a uma temperatura de transição escolhida, o que pode torná-los mais adequados para janelas estreitas, como 2-8°C ou transporte ambiente controlado. No transporte de biotecnologia, esse comportamento da fase é valioso porque pode reduzir o choque frio durante as primeiras horas após a embalagem. Um pacote que muda de fase perto do intervalo alvo pode absorver calor sem conduzir a carga útil para baixo em direção ao ponto de congelamento, da mesma forma que um pacote à base de água totalmente congelado às vezes pode..
O desempenho térmico ainda depende da montagem mais ampla. Espessura da embalagem, massa total de PCM, a distância entre o refrigerante e o produto, e a massa térmica da carga influenciam quanto tempo a remessa permanece dentro do alcance. Até o protocolo de condicionamento é importante. Um fornecedor deve ser capaz de explicar se a embalagem se destina a ser utilizada totalmente acondicionada, parcialmente condicionado, ou em combinação com outros componentes.
Em operações de biotecnologia, Os pacotes PCM são normalmente considerados para distribuição refrigerada de reagentes, remessas de amostras de ensaios clínicos, e transporte de kit de diagnóstico onde as excursões térmicas podem invalidar os resultados. Cada caso tem seu próprio perfil de sensibilidade. Alguns reagentes não devem congelar. Algumas amostras precisam de um ambiente refrigerado estável durante o transporte. Alguns kits contêm vários componentes com diferentes tolerâncias térmicas, o que significa que o pack-out deve proteger o elemento mais sensível e não o mais fácil.
Onde os compradores ganham valor e onde começam os erros
Pacotes PCM podem reduzir o excesso de temperatura, suporta janelas de controle mais estreitas, e tornar as embalagens refrigeradas mais tolerantes quando o produto deve ficar longe de temperaturas congelantes. Eles são especialmente atraentes quando o custo da perda de carga útil é alto ou quando o laboratório receptor não pode tolerar muita variação.
Mas PCM não é mágica. Pode ser mais caro, pode exigir um condicionamento mais disciplinado, e a temperatura da fase escolhida deve corresponder aos verdadeiros requisitos do produto, em vez de uma suposição esperançosa. Um PCM de 5°C não garante uma remessa de 2 a 8°C, a menos que o restante da configuração suporte isso. Da mesma maneira, uma janela térmica estreita só é útil se a carga útil, isolamento, e a rota são estáveis o suficiente para que essa precisão seja importante.
Para aquisição de PCM, pergunte o que a temperatura de fase declarada realmente significa na prática. É um ponto de transição nominal, uma faixa testada, ou uma etiqueta de marketing? Pergunte como o pacote está condicionado, quanto tempo deve equilibrar antes da embalagem, e como o posicionamento em torno da carga afeta o resfriamento nas primeiras horas. Essas perguntas muitas vezes revelam se o fornecedor entende de manipulação biotecnológica ou está apenas revendendo um pacote genérico de fases..
Os modos de falha geralmente aparecem nos limites: condicionamento incompleto, contato direto com um frasco sensível ao congelamento, muito pouca massa de PCM para o tamanho da carga útil, ou muita massa para uma remessa pequena que fica mais fria do que o esperado. O objetivo da revisão técnica não é complicar a compra. É tornar visíveis esses erros previsíveis antes da expansão.
Uma lista prática de fornecedores
Para compras de biotecnologia, a auditoria deve concentrar-se nas evidências e na repetibilidade, e não na linguagem de marketing. Os compradores de biotecnologia geralmente compram PCM porque a janela de remessa é estreita e a carga útil é muito valiosa para suposições. Reagentes, kits de ensaio, amostras clínicas, enzimas, e alguns materiais baseados em células podem precisar de um controle mais rígido do que um pacote genérico de gel congelado pode oferecer.
A lista certa baseia-se na repetibilidade, ajustar, e limites operacionais honestos. Peça ao fornecedor para responder aos pontos abaixo por escrito para que a aprovação da amostra e a aprovação em massa fiquem alinhadas.
Confirme as dimensões internas e externas, preencher peso, e quantidades de caixas para que o pacote se ajuste ao seu remetente atual sem desperdício de espaço aéreo.
Pergunte qual filme ou material externo é usado, como os selos são formados, e quais controles existem para evitar desvios entre lotes.
Solicite instruções de condicionamento por escrito em vez de depender de hábitos informais de congelamento na bancada de embalagem.
Verifique se os pacotes de amostras e os pacotes de produção vêm da mesma lista de materiais, a mesma rotina de preenchimento, e o mesmo padrão de qualidade.
Pergunte como o fornecedor comunica qualquer formulação, filme, imprimir, ou mudança de dimensão do pacote antes do envio.
Temperatura exata de mudança de fase e se o fornecedor oferece vários pontos de ajuste
Como o pacote é condicionado, reiniciar, e embalado para evitar choques frios indesejados
Construção de filme, resistência a vazamentos, e consistência de preenchimento lote a lote
Processo de controle de alterações para resina, formulação de gel, ou mudanças na linha de fabricação
Suporte para testes piloto, revisão do registrador, e correspondência entre amostra e produção
Esclareça se o pacote se destina a ser um componente de um expedidor qualificado ou simplesmente um refrigerante geral para uso mais amplo.
Execute um pequeno piloto com um registrador antes de dimensionar. Um fornecedor confiável deve se sentir confortável em apoiar essa etapa.
Como validar antes de dimensionar
Antes de um grande pedido, uma corrida piloto vale o tempo. Use pacotes com intenção de produção no remetente isolado exato, com massa de carga útil real, prática de condicionamento real, e um registrador. Esse pequeno exercício muitas vezes revela se o problema é a escolha do refrigerante, colocação do pacote, rotina do freezer, ajuste da caixa, ou recebendo disciplina. Registre não apenas o rastreamento do logger, mas também a temperatura de carregamento do produto, o número exato e a localização dos pacotes, o tempo que a caixa ficou aberta durante a embalagem, e as condições ambientais no momento da expedição.
Depois do piloto, revisar mais do que passar/reprovar. Procure por picos de frio, aquecimento tardio, condensação, quebra de pacote, e lidar com o atrito. Muitas equipes descobrem que o principal problema não era a química do gel; foi a colocação do pacote, Tamanho da caixa, rotina do freezer, ou uma incompatibilidade entre o pacote de amostras e o fornecimento com intenção de produção.
Pergunte aos fornecedores quais dados eles podem fornecer e quais não podem. Um parceiro responsável pode discutir as dimensões da embalagem, ponto de fase, e condições gerais de uso, mas não deve implicar que um pacote PCM valide automaticamente sua pista. Se o seu produto estiver sob controle do estilo GDP ou sob um protocolo de estudo, qualificação adicional pode ser necessária.
O segredo é fazer com que o fornecedor prove que a embalagem cotada pode ser fabricada e acondicionada sempre da mesma forma.. Em trabalhos laboratoriais sensíveis, a reprodutibilidade é muitas vezes o fator de compra decisivo.
Para onde estão indo as atuais prioridades de sourcing
A direção do mercado em biotecnologia é em direção a um controle de temperatura mais restrito, melhor uso do registrador, e menos pacotes de tamanho único. Os atacadistas preferem cada vez mais fornecedores que possam apoiar corridas piloto, revisão de dados, e repetir o controle de fabricação em vez de simples transações de catálogo. A sustentabilidade também faz parte da discussão, mas os compradores de biotecnologia geralmente colocam a confiabilidade térmica em primeiro lugar. A estratégia de sustentabilidade mais realista não consiste simplesmente em utilizar menos materiais. É para reduzir a perda de carga útil, evitar remessas repetidas, e escolha pacotes que se ajustem ao sistema isolado de forma eficiente, para que o desperdício e o peso dimensional sejam controlados.
O fornecimento no atacado também está avançando em direção a uma melhor documentação. Os compradores pedem cada vez mais detalhes da mudança de fase, rotinas de condicionamento recomendadas, e evidência de que um lote de substituição se comportará como o lote aprovado. Essa mudança favorece os fornecedores com disciplina de processo em detrimento dos comerciantes que apenas encaminham fichas de catálogo.
Conclusão
No transporte de biotecnologia, a decisão vencedora raramente é a fonte de frio mais barata ou a ficha de catálogo mais técnica. É a matilha que apoia um disciplinado, repetível, embalagem baseada em evidências para o produto real e rota real.
Isso geralmente significa escolher o fornecedor que entende os limites de qualificação, disciplina de condicionamento, e controle lote a lote tão claramente quanto entendem o próprio PCM.
Sobre Huizhou
Em Huizhou, nos concentramos em embalagens com temperatura controlada para cadeia de frio para alimentos, medicamento, e outras remessas sensíveis à temperatura. Nossa linha de produtos listada publicamente inclui bolsas de gelo em gel, tijolos de gelo congelador, forros de caixa isolados, Caixas EPP, Tampas de paletes, e materiais de embalagem relacionados. Também descrevemos nosso trabalho em torno do desenvolvimento de soluções de cadeia de frio com R interno&D e suporte para testes térmicos. Isso nos ajuda a discutir tanto os refrigerantes individuais quanto o sistema de embalagem mais amplo ao seu redor.
Próximo passo
Se você estiver revisando fornecedores ou planejando uma nova embalagem, comece com a faixa real de temperatura do produto e o comprimento da rota. Em seguida, peça um conjunto de amostras que corresponda ao seu pedido em massa pretendido e teste-o antes de dimensionar.
Perguntas frequentes
Estas são as perguntas que mais frequentemente permanecem após o término da comparação inicial.
Por que os compradores de biotecnologia escolhem o PCM em vez do gel congelado comum?
Porque o PCM pode reter energia térmica em torno de um ponto de transição escolhido. Isso torna mais fácil projetar uma remessa que proteja uma gama restrita de produtos, em vez de simplesmente deixar a embalagem o mais fria possível.. Se a carga útil for sensível ao congelamento, valide a resposta com uma avaliação do logger antes de aprovar o uso rotineiro.
Um pacote de gel PCM pode substituir uma caixa de remessa isolada?
Não. A caixa, isolamento, padrão de carregamento de carga útil, e o perfil da rota determinam o desempenho no mundo real. O PCM ajuda apenas quando o restante do sistema corresponde ao produto e à pista. Uma janela estreita de temperatura só é útil quando o empacotamento em torno do PCM é igualmente disciplinado.
Quais dados um atacadista deve solicitar antes de aprovar um fornecedor?
Solicite a temperatura alvo da fase, instruções de condicionamento, peso nominal ou tolerância de preenchimento, construção de filme externo, tempo de redefinição, e pacotes de amostras para testes de rota. Para aprovação no atacado, confirme a resposta em relação à sua carga útil real, remetente, e método de condicionamento.































