Pharma cold chain services keep temperaturesensitive medicines safe from manufacturer to patient. Sem uma logística confiável da cadeia de frio, up to half of all vaccines are wasted due to poor temperature control. Recent growth in biologics, gene therapies and weightloss drugs has increased demand for ultracold and refrigerated distribution. Em 2020 temperaturecontrolled logistics already accounted for nearly 18 % dos gastos com logística biofarmacêutica, and that share is rising. This article explains what makes pharma cold chain services unique, por que eles são importantes para você, and how new technologies and regulations shape the industry in 2025.
O que são cadeia de frio farmacêutica serviços? A clear definition and overview of temperaturecontrolled logistics for medicines.
Why does the cold chain matter for biologics and vaccines? Learn about temperature ranges, product sensitivity and waste prevention.
Which market trends drive demand? Discover how GLP1 therapies and gene treatments are expanding the cold chain market.
What technologies and innovations shape 2025? Explore sensores IoT, Otimização de rotas de IA, blockchain, sustainable power and automation.
How do regulations and best practices ensure compliance? Entenda as Boas Práticas de Distribuição (PIB), WHO guidelines and quality management.
How to choose a cold chain partner? Practical tips and criteria for selecting a service provider.
What Are Pharma Cold Chain Services and Why Do They Matter?
Pharma cold chain services refer to the endtoend logistics processes that keep medicines within specific temperature ranges during storage, embalagem e transporte. Temperature control preserves the potency and safety of drugs; failures can render vaccines or biologics ineffective, causing public health risks. Aproximadamente 50 % of vaccines are wasted each year due to inadequate temperature management, illustrating the stakes. Pharma cold chain services encompass refrigerated (2 °C8 °C), congelado (–20 °C a –80 °C) e criogênico (abaixo de –80 °C) armazenar, along with roomtemperature control (20 °C25 °C) para determinados produtos.
Understanding Temperature Sensitivity Across Therapies
Biologics and biosimilars dominate new drug approvals—every third new medicine is a biologic—and more than 85 % of these drugs require cold chain management. Vaccines are among the most temperaturesensitive products: mRNA vaccines for COVID19 must be stored between –60 °C and –80 °C, Moderna’s mRNA vaccine at –20 °C, and traditional vaccines at 2 °C8 °C. Advanced cell and gene therapies need cryogenic storage at –150 °C or lower, while peptides like GLP1 agonists for diabetes and obesity rely on refrigerated conditions. These varied requirements mean cold chain services must offer multiple temperature zones and precision control to avoid degradation.
| Faixa de temperatura | Produtos de exemplo | Significado prático |
| 2 °C–8 °C (refrigerado) | Vacinas tradicionais, Anticorpos monoclonais, GLP1 agonists | Most drugs fall into this range; maintaining refrigeration prevents protein denaturation and ensures stability. |
| –20 °C a –80 °C (frozen/deepfrozen) | Moderna mRNA vaccine (–20 °C), weightloss injectables, certain enzymes | Requires insulated packaging with phasechange materials or dry ice for long transit durations. |
| Abaixo de –80 °C (criogênico) | Terapias celulares e genéticas, CART cell treatments | Demands specialised freezers, liquid nitrogen vapor storage and robust monitoring to protect living cells. |
| 20 °C–25 °C (sala controlada) | Smallmolecule drugs, tablets and capsules | Though less demanding, stable ambient control prevents humidity or heat damage. |
Dicas e conselhos práticos
Know your product’s temperature profile: Different drugs have unique stability data. Request stability studies from manufacturers to set appropriate temperature bands.
Use embalagens validadas: Choose containers qualified for your product’s thermal requirements, such as batterypowered active containers for long transit or insulated expedidores passivos for short routes.
Planeje rotas com cuidado: Factor in climate zones, customs delays and transfer points. Work with carriers experienced in cold chain logistics.
Monitore continuamente: Deploy IoTenabled sensors or data loggers to record temperature, umidade e localização. Realtime monitoring allows intervention before excursions occur.
Treine sua equipe: Educar a equipe sobre os procedimentos de manuseio, lendo registros de temperatura e respondendo a alarmes. Human error contributes significantly to excursions.
Caso real: Durante o lançamento da vacina COVID19, several shipments were rejected because data loggers showed brief temperature excursions during airport transfers. By preconditioning shippers longer and adding gelo seco at layovers, the logistics team eliminated excursions and saved thousands of doses.
How Do Logistics and Storage Maintain Temperature Integrity?
Cold chain logistics maintain temperature integrity through specialised packaging, storage infrastructure and qualified transportation. Warehouses provide multiple zones: refrigerated suites, freezers and cryogenic rooms for ultracold storage. Temperaturecontrolled trucks and aircraft maintain product conditions during transit. Realtime monitoring and alarm systems ensure any deviation is detected immediately. Por exemplo, global health authorities emphasise continuous monitoring in guidelines, and logistic providers report that temperature integrity is the top selection criterion for 44 % of customers.
Storage and Transport Methods: Active vs Passive Solutions
There are two primary methods for keeping products cold during transport:
Soluções ativas usar refrigeração elétrica (baterias, compressores) para manter temperaturas precisas. These units, such as Envirotainer’s RKN and RAP containers, can operate for 130–170 hours at 4 °C–30 °C and include sensors for condition monitoring. Active containers suit longdistance shipments and highvalue biologics.
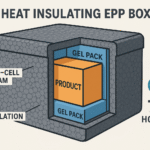
Soluções passivas rely on insulation and phasechange materials (pacotes de gel, gelo seco). They include Tampas de paletes like Sonoco ThermoSafe’s Pegasus ULD, which offers up to 300 horas de 2 Serviço °C–8 °C. Passive systems are lighter and simpler but may require reicing for extended journeys.
Storage facilities also differentiate between sala controlada, refrigerado, congelado e criogênico zonas. Modern warehouses integrate insulated panels, redundant refrigeration units and backup power. Inventory management systems track batch location, expiration dates and temperature history, ensuring chainofcustody documentation.
| Storage/transport method | Características | Melhor caso de uso | Considerações |
| Recipiente refrigerado ativo | Onboard battery or engine; compressors regulate temperature; sensors and telemetry | Longhaul flights or transcontinental shipping of highvalue biologics | Heavier and costlier; requires charging and maintenance |
| Remetente isolado passivo | Multilayer insulation, materiais de mudança de fase, gelo seco; no power required | Short to midrange shipments, lastmile delivery | Duração limitada; weight limitations; reicing needed on long trips |
| Cryogenic shipping system | Uses liquid nitrogen or dry vapor to maintain –150 °C for cell therapies | Transport of CART cells, amostras de tecido, terapias genéticas | Strict safety protocols; specialised handling and training |
| Portable freezer or refrigerator | Small units for clinics or field use; solarpowered options emerging | Rural vaccination campaigns, disaster relief missions | Must ensure consistent power supply or solar charging |
Dicas e conselhos práticos
Equilibre custo e risco: Use active containers for highvalue or ultrasensitive products; passive shippers may suffice for stable vaccines and peptides.
Validar pistas: Test packaging and routes under worstcase conditions to ensure they maintain temperature across possible delays.
Manter o equipamento: Faça a manutenção regular das unidades de refrigeração, freezers e sensores. Battery failures are a common cause of excursions.
Estabeleça planos de contingência: Prepare for unexpected delays, customs holds or weather disruptions by having spare packaging, extra gel packs and local storage options.
Documente tudo: Regulatory inspections require temperature logs, maintenance records and deviation reports.
Exemplo real: A contract manufacturer shipping monoclonal antibody vials from Europe to the US used passive shippers with phasechange materials. When winter storms delayed flights, the gel packs’ cooling capacity was exhausted. By switching to active containers with battery backup, a empresa manteve 2 °C–8 °C para 96 hours and prevented product loss.
Which Technologies and Innovations Are Transforming Pharma Cold Chain Services?
Technological innovation is reshaping cold chain logistics, enabling realtime control, predictive insights and sustainable operations. Automation and robotics are reducing labor costs and errors; aproximadamente 80 % dos armazéns ainda carecem de automação, indicando potencial de crescimento significativo. A sustentabilidade é agora um valor fundamental: energyefficient refrigeration, recyclable packaging and lowemission transportation are essential. Endtoend visibility through IoT tracking devices provides location and temperature data, permitindo otimização e conformidade de rotas.
Inovações emergentes: IoT, IA, Blockchain and Sustainable Power
Several technological trends to watch in 2025 incluir:
IoTenabled sensors and realtime tracking – Hardware comprises over 76 % do mercado de rastreamento da cadeia de frio. Sensores registram temperatura, umidade e choques, enviando alertas quando as condições divergem. This data enables proactive interventions and supports regulatory audits. Integrating GPS ensures route optimisation and lastmile accuracy.
Inteligência artificial (IA) e análise preditiva – AI forecasts demand, identifies potential equipment failures and optimises routes based on traffic and weather. AIdriven demand forecasting helps allocate inventory strategically, reduzindo a deterioração.
Blockchain para rastreabilidade ponta a ponta – Distributed ledger technology records each step in the cold chain, preventing data tampering and ensuring regulatory compliance. Blockchain improves trust between manufacturers, logistics providers and regulators by making transaction histories immutable.
Solarpowered storage and sustainable refrigeration – Solar armazenamento refrigerado units reduce energy costs and provide reliable power in rural areas, addressing inconsistent grids. Green fuels and renewable energy are increasingly used in refrigerated trucks and warehouses.
Congeladores criogênicos portáteis – New portable freezers maintain –80 °C to –150 °C for cell therapies and gene treatments. These units ensure ultracold conditions without heavy infrastructure, facilitating clinical trials and personalized therapies.
Embalagens e sensores inteligentes – Companies like World Courier embed smart sensors in multiuse packaging to enable customers to track shipments through a portal, delivering data on location, temperatura e choques.
Sistemas automatizados de armazenamento e recuperação (AS/RS) – Robots and automated conveyors handle pallet movements in cold warehouses, improving throughput and reducing human exposure to cold environments.
| Inovação | Descrição | Benefício para você |
| Sensores de IoT | Sensores monitoram a temperatura, umidade e localização; hardware segment accounted for over 76 % do mercado de rastreamento | Allows realtime visibility and quicker response to excursions, evitando a deterioração. |
| Análise de IA | Algoritmos prevêem demanda, predict equipment failure and optimise routes | Reduces costs and delays; improves resource allocation and maintenance planning. |
| Blockchain | O razão distribuído registra cada transação; ensures tamperproof documentation | Enhances compliance and trust by providing an immutable record of product conditions and chain of custody. |
| Energia solar | Solarpowered cold storage units reduce operational costs and enable rural distribution | Cuts energy expenditure and supports sustainable supply chains, especialmente em áreas remotas. |
| Congeladores criogênicos | Portable freezers maintain –80 °C to –150 °C for cell therapies | Enables decentralised clinical trials and personalized therapy distribution. |
| Embalagem inteligente | Integrated sensors transmit data to networks for proactive intervention | Provides detailed shipment status to carriers and clients, enhancing transparency and decisionmaking. |
Dicas e conselhos práticos
Invest in technology that matches your scale: Start with data loggers and GPS trackers; scale up to AIdriven analytics as shipment volumes grow.
Priorize a segurança cibernética: Protect IoT and blockchain platforms from hacking or data tampering; use encryption and regular audits.
Abrace a sustentabilidade: Choose refrigeration units with lower power consumption; consider renewable energy and recyclable packaging to meet ESG goals.
Pilot emerging innovations: Test portable cryogenic freezers or solarpowered storage in controlled trials before full deployment.
Colabore com fornecedores de tecnologia: Work with vendors experienced in pharmaceutical regulations to ensure compliance from design to implementation.
Insight de caso: A logistics provider implemented AI route optimisation to deliver GLP1 peptides to remote pharmacies. The system combined realtime traffic data and weather forecasts, reducing average delivery time by 15 % and preventing temperature deviations. A satisfação do cliente melhorou, and waste decreased.
What Are the Regulatory and Compliance Requirements for Pharmaceutical Cold Chains?
Strict regulations govern pharmaceutical cold chain operations to protect patient safety and maintain drug efficacy. Authorities such as the U.S. Food and Drug Administration (FDA), Agência Europeia de Medicamentos (Ema) e Organização Mundial da Saúde (QUEM) aplicar Boas Práticas de Fabricação (GMP) e Boas Práticas de Distribuição (PIB) diretrizes. These guidelines mandate validated temperature control, documentação, quality management and traceability throughout the supply chain. Noncompliance can lead to product recalls, multas e danos à reputação.
Navigating GDP, GMP and Regulatory Guidelines
Key regulatory concepts include:
Boas Práticas de Distribuição (PIB): Defines standards for procurement, armazenamento e transporte. GDP requires riskbased temperature mapping of storage areas, qualification of vehicles and equipment, robust quality management systems and proper documentation. Regulators emphasise realtime monitoring and data integrity to ensure products remain within specified ranges.
Boas Práticas de Fabricação (GMP): Covers manufacturing environments. Para cadeia de frio, GMP requires validated processes for temperature control during production, aseptic filling and packaging.
Diretrizes da OMS para armazenamento de vacinas: Standardize conditions for vaccines from 2 °C–8 °C for refrigerated products to –50 °C–15 °C for frozen vaccines; they highlight that failure to maintain these ranges contributes to 50 % desperdício de vacina.
Serialization and anticounterfeiting: Many jurisdictions require unique identifiers on packaging and tamperevident seals. Blockchain is increasingly used to ensure authenticity and traceability.
Mapeamento e qualificação de temperatura: Storage and transport units must be mapped under worstcase conditions to prove they maintain required temperatures. Calibration of sensors and data loggers must be uptodate.
| Guideline or regulation | Requisitos principais | Implicações |
| PIB | Qualified vehicles, mapeamento de temperatura, documented SOPs and training | Ensures consistent distribution practices; failing to map temperatures can lead to noncompliance. |
| GMP | Validated manufacturing processes, controle ambiental, aseptic filling | Protects product integrity during production; requires continuous monitoring and documented protocols. |
| Diretrizes de vacinas da OMS | 2 °C–8 °C para a maioria das vacinas; –50 °C–15 °C for frozen; emphasises 50 % vaccine wastage due to poor cold chain | Highlights global need for improved infrastructure and management; influences national immunisation programmes. |
| Serialização | Unique identifiers on packaging; data integrity across supply chain | Prevents counterfeiting and enables recall traceability; requires digital systems and compliance with global standards. |
| Mapeamento de temperatura | Qualify storage and transport units under worstcase scenarios | Provides evidence to regulators; supports risk assessment and contingency planning. |
Dicas e conselhos práticos
Implementar um sistema de gestão da qualidade: Documentar POPs, training records and deviation management procedures. Assign a responsible person for GDP compliance.
Calibrar regularmente o equipamento: Verify that sensors and data loggers are accurate; maintain calibration certificates for inspections.
Realizar auditorias simuladas: Simulate regulatory inspections to identify gaps and train staff on compliance.
Acompanhe atualizações regulatórias: Monitor changes in FDA, EMA and WHO guidance; adjust protocols accordingly.
Use tamperevident and serialised packaging: Helps prevent counterfeit infiltration and ensures traceability.
Compliance case: Durante uma inspeção do PIB, a UK distributor failed to demonstrate supplier qualification and proper temperature mapping, resulting in a major deficiency report. After implementing a riskbased mapping programme and upgrading its documentation system, the company successfully passed a followup audit.
Choosing the Right Pharma Cold Chain Partner: Considerations for 2025
Selecting a cold chain partner involves assessing experience, infraestrutura, práticas de tecnologia e sustentabilidade. The market for pharmaceutical logistics services was estimated at USD 99.33 bilhão em 2024 e está projetado para atingir USD 208.26 bilhão por 2033 com um CAGR de 8.5 %. This growth indicates intense competition among service providers and abundant options for shippers.
Evaluating Providers: Capacidade, Tecnologia e Sustentabilidade
Ao escolher um parceiro, considere os seguintes critérios:
Experience with similar products: Look for providers who handle biologics, vaccines or cell therapies and have proven capability across required temperature ranges.
Infrastructure and global reach: Assess warehouse capacity, number of temperaturecontrolled vehicles and geographic coverage. Multicontinent networks reduce transit time and risk.
Adoção de tecnologia: Determine whether the provider uses IoT sensors, realtime tracking, AI analytics and integrated systems for inventory and documentation.
Conformidade regulatória: Check for GDP/GMP certifications, quality audits and documented processes. An accredited quality management system signals maturity.
Compromisso de sustentabilidade: Consider providers investing in reusable packaging, renewable energy and emissionreduction initiatives.
Financial stability: Evaluate track record, investment in capacity expansion and ability to invest in future technologies.
| Evaluation criterion | O que procurar | Por que isso importa |
| Therapeutic expertise | Experience with vaccines, Biologics, peptídeos, terapias celulares | Ensures the provider understands temperature requirements and regulatory demands. |
| Infrastructure scale | Várias zonas de temperatura; cryogenic capabilities; rede global | Supports efficient distribution and reduces risk of excursions during long journeys. |
| Tecnologia | Sensores de IoT, IA, blockchain, armazéns automatizados | Melhora a visibilidade, reduces errors and improves efficiency. |
| Conformidade | GDP/GMP certifications; histórico de auditoria | Reduces regulatory risk and demonstrates quality commitment. |
| Sustentabilidade | Embalagem reutilizável; green power; emission targets | Aligns with ESG goals and reduces environmental impact. |
| Suporte ao cliente | 24/7 monitoramento; Gerenciamento de conta dedicado; treinamento | Ensures responsive service and problem resolution. |
Dicas e conselhos práticos
Solicite estudos de caso: Ask potential partners for examples of similar shipments and performance metrics (entrega dentro do prazo, taxa de excursão). Review how they handled disruptions.
Visite instalações: Tour warehouses to inspect cleanliness, organisation and temperature control measures. Confirm backup power and contingency plans.
Evaluate data integration: Ensure your IT systems can interface with the provider’s tracking and documentation platforms; look for open APIs.
Negotiate clear SLAs: Definir faixas de temperatura, response times for alarms, reporting frequency and remedies for excursions.
Teste antes de dimensionar: Conduct pilot shipments to verify performance and gather data before committing large volumes.
Real world scenario: A biotech firm evaluated two cold chain providers. One offered lower rates but lacked cryogenic capacity and realtime monitoring. The other had robust infrastructure, AIdriven analytics and certified GDP procedures. After pilot testing, the biotech chose the latter, resulting in fewer temperature deviations and faster regulatory approvals.
2025 Latest Developments and Trends in Pharma Cold Chain Services
Visão geral da tendência
The pharma cold chain landscape continues to evolve in 2025. O mercado global de logística da cadeia de frio na área da saúde, avaliado em USD 59.97 bilhão em 2024, está projetado para atingir USD 65.14 bilhão em 2025 e dólares americanos 137.13 bilhão por 2034, crescendo em um CAGR de 8.63 %. Rapid growth is driven by rising demand for temperaturesensitive biologics, vaccines and blood products. North America remains dominant due to its large biopharma industry, while AsiaPacific records the fastest growth thanks to expanding vaccine manufacturing and healthcare investment. The pharmaceutical logistics market more broadly is expected to reach USD 208.26 bilhão por 2033.
Instantâneo dos últimos avanços
Weightloss and diabetes biologics boom: The popularity of GLP1 receptor agonists such as Ozempic, Wegovy and Mounjaro has surged, reaching sales of $40.6 billion for Novo Nordisk and $5.4 billion for Eli Lilly in 2024. These drugs require refrigerated storage and drive expansion of cold chain capacity.
Cell and gene therapies scale up: Although cell and gene therapies represent only 0.2 % of prescription volume, they account for 32 % of industry revenue and require cryogenic logistics. Investments in cryogenic contêineres de transporte and specialised handling are increasing.
Investimento e consolidação: Private equity interest remains high; companies like Frontier Science Solutions and MD Logistics are expanding facilities across North America. New batterypowered reusable containers (por exemplo, MedStow Micro) and cryogenic carriers (Cryoport Express HV3) offer extended service durations.
Regulatory focus on traceability and data integrity: Blockchain and AI are being integrated into supply chains to enhance transparency and comply with tightening GDP and GMP requirements.
Iniciativas de sustentabilidade: Operators are adopting solar power, loweremission refrigeration and recyclable packaging; global food cold chain infrastructure accounts for ~2 % das emissões de CO₂, prompting greener strategies.
Mercados emergentes: Southeast Asia is a hotbed for innovations like solarpowered storage, blockchain tracking and portable cryogenic freezers.
Insights de mercado
Consumer preference for fresh products and online grocery shopping is increasing demand for cold chain logistics. The North American cold chain market is projected to grow from USD 116.85 bilhão em 2024 para USD 289.58 bilhão por 2034 em um CAGR de 9.5 %, with expansion driven by pharma, ecommerce and sustainability practices. Rising environmental consciousness is prompting companies to adopt ecofriendly packaging and energyefficient refrigeration. Investimento em novas instalações, such as CJ Logistics’ cold storage near Kansas City and Lineage Logistics’ crossborder network, reflects this growth.
Perguntas frequentes
Q1: What are the main temperature ranges for pharmaceutical cold chain products?
Most pharmaceuticals fall into four temperature categories: temperatura ambiente controlada (20 °C–25 °C), refrigerado (2 °C–8 °C), frozen/deepfrozen (–20 °C a –80 °C) e criogênico (abaixo de –80 °C). Each range suits different therapies—vaccines and monoclonal antibodies require 2 °C–8 °C, while cell and gene therapies need cryogenic storage. Knowing your product’s required range ensures you select appropriate packaging and transport.
Q2: Why is the cold chain market growing so quickly?
Demanda por produtos biológicos, vacinas, peptides and personalized medicines is surging. The healthcare cold chain logistics market grew from USD 59.97 bilhão em 2024 e está projetado para atingir USD 137.13 bilhão por 2034. Weightloss biologics and cell therapies drive significant capacity expansion.
Q3: Como os sensores IoT melhoram o gerenciamento da cadeia de frio?
Dispositivos IoT monitoram a temperatura, umidade e localização em tempo real. They allow immediate intervention if a shipment deviates from its temperature range and provide digital records for regulatory compliance. Hardware represented over 76 % do mercado de rastreamento da cadeia de frio em 2022.
Q4: What is the difference between active and embalagem passiva da cadeia de frio?
Active packaging uses powered refrigeration and can maintain precise temperatures for longer durations; examples include batterypowered containers with sensors. Passive packaging relies on insulation and phasechange materials like gel packs or dry ice. Active systems suit long journeys and highvalue biologics, while passive systems are lighter and costeffective for shorter routes.
Q5: What should I look for when choosing a cold chain service provider?
Assess the provider’s experience with your product type, infrastructure scale, adoção de tecnologia, regulatory compliance and sustainability practices. Peça estudos de caso, visit facilities and test small shipments before scaling up.
Resumo e sugestões
Resumindo, pharma cold chain services preserve lifesaving medicines by maintaining specific temperatures across storage, embalagem e transporte. Com quase 18 % of biopharma logistics spending devoted to temperaturecontrolled logistics and 85 % of biologics requiring cold chain management, the importance of these services cannot be overstated. Market growth is robust—healthcare cold chain logistics are expected to reach USD 137.13 billion by 2034—driven by biologics, vacinas, GLP1 therapies and gene treatments. Novas tecnologias, como sensores IoT, Análise de IA, blockchain and sustainable energy improve visibility, reduza o desperdício e apoie a conformidade regulatória. Para ter sucesso, companies should invest in modern equipment, treinar pessoal, validate routes and adopt a quality management culture.
Recomendações de ação
Audite suas necessidades de cadeia de frio: Assess each product’s temperature range, sensitivity and shelf life to inform packaging and transport choices.
Strengthen monitoring: Implement IoTbased sensors and realtime tracking to gain visibility and compliance.
Invista em soluções sustentáveis: Adopt energyefficient refrigeration and recyclable packaging to meet environmental commitments and reduce operational costs.
Faça parceria com sabedoria: Choose providers with proven experience, robust infrastructure and advanced technology; require clear service level agreements and conduct pilot shipments.
Mantenha-se informado: Follow regulatory updates and industry trends to anticipate new requirements and leverage emerging innovations.
Sobre Tempk
Tempk is a leading provider of integrated soluções de cadeia fria for the pharmaceutical and life sciences industry. Our team combines decades of experience with cuttingedge technology to deliver reliable, endtoend temperaturecontrolled logistics. We offer multitemperature storage, ativo e passivo soluções de embalagem, and realtime monitoring systems to ensure product integrity. Our sustainable initiatives include reusable packaging, solarpowered facilities and route optimisation algorithms, reducing carbon footprint while maintaining compliance. With a global network of GDPcertified sites and a customercentric approach, we help you bring lifesaving medicines to patients safely and efficiently.
Chamado à ação: Contact Tempk’s experts to discuss your cold chain challenges and learn how our tailored solutions can protect your products and support your growth. Junto, we ensure temperature integrity from manufacture to administration.