Vaccine Ice Box Vendor Price
A buyer searching for vaccine ice box vendor price is usually trying to solve a practical problem: how to obtain a reliable insulated container without overpaying for the wrong construction or underbuying the wrong performance. That is especially true in cold-chain packaging, where a ice box may look simple but still sits inside a larger operating system. The box must match the payload, the refrigerant, l'itinéraire, the handling pattern, and the commercial model. A vendor often sits closer to the commercial interface than the factory floor. That can help with quotations and sourcing convenience, but buyers should verify build origin, technical ownership, and what support exists after the PO is issued. This matters because these containers are commonly used across last-mile immunization transport, outreach clinics, temporary transfer between facilities, emergency relocation of stock, and other movements of temperature-sensitive biologics that cannot tolerate casual handling. The most expensive mistake is to treat a vaccine ice box as a generic cooler. Vaccine transport is part of a cold chain, and cold chain failure can reduce potency even when the container still feels cold to the touch.
What the market usually means by Vaccine Ice Box Vendor Price
In sourcing language, terms like manufacturer, provider, vendor, distributeur, usine, or company are not interchangeable. They shape what kind of control sits behind the quote. A manufacturer or factory may control tooling, moulage, construction de mur, assemblée, and release standards. A distributor may offer stocked models and faster availability but less freedom to change dimensions or add technical documentation. A provider or vendor may coordinate several upstream sources and bundle more services, but you still need to know which plant actually makes the product.
A vendor often sits closer to the commercial interface than the factory floor. That can help with quotations and sourcing convenience, but buyers should verify build origin, technical ownership, and what support exists after the PO is issued.
A realistic quotation for vaccine ice box vendor price should separate base product cost from system cost. Base product cost covers the physical box. System cost covers the box plus refrigerant, fardage, instructions de travail, surveillance, packaging labor, and any testing or qualification work needed to prove the packout. Buyers who ask for that separation usually make faster and safer decisions.
Because the search intent here is clearly commercial, the most useful first output from any supplier is a structured quotation. Ask for the empty ice box specification, the insulation description, the minimum order logic, the accessory list, and the freight basis. If customization is involved, separate one-time tooling from repeat unit price. Si l'envoi est sensible à la température, separate the container price from the full packout price. That keeps negotiations honest and helps your internal team compare offers on the same assumptions.
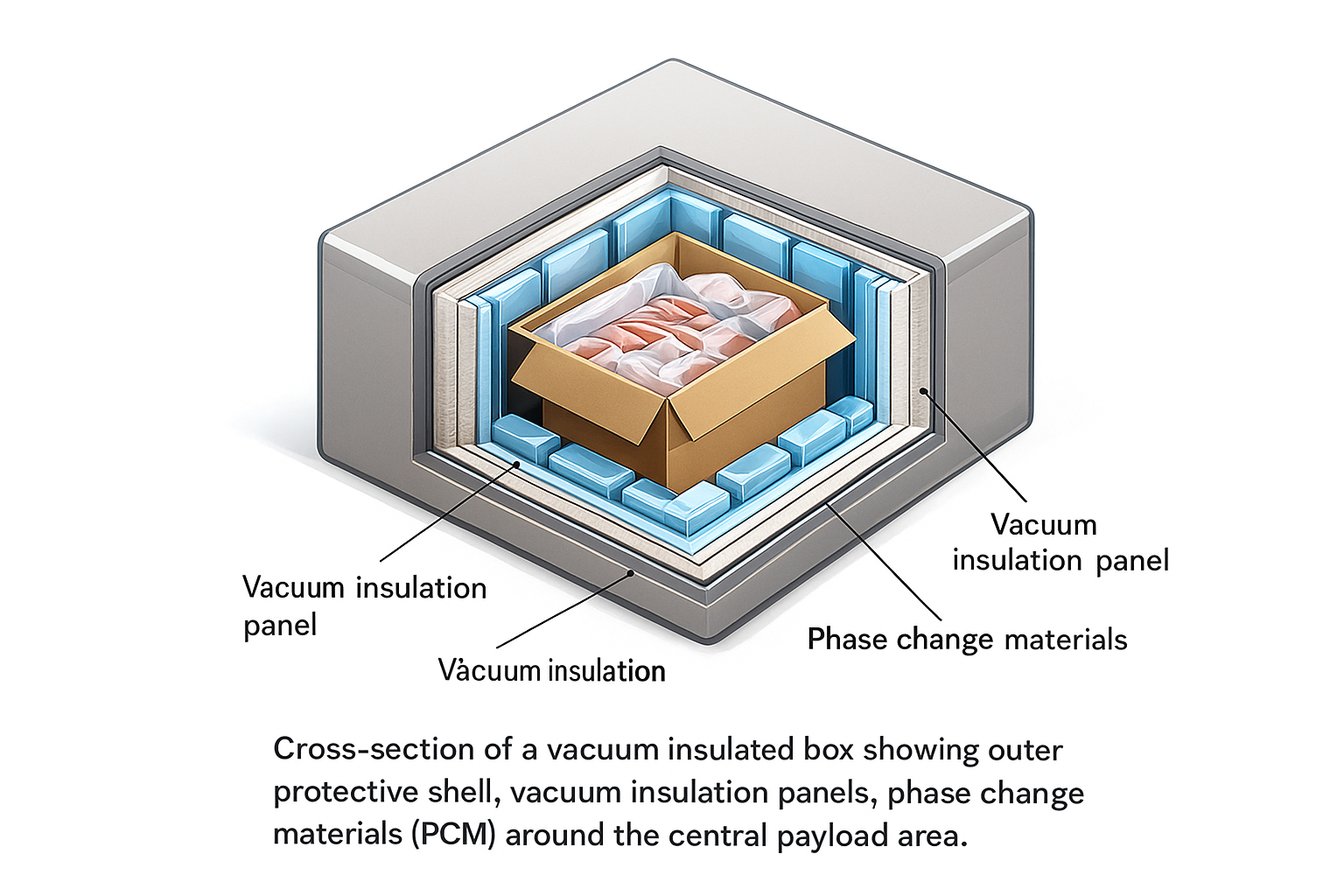
Matériels, construction, and why they change the quote
Most buyers see an insulated ice box as one product. En réalité, it is usually a layered construction with at least three decisions behind it: the outer structure, the insulation strategy, and the coolant interface. Those three choices drive durability, temperature margin, poids, hygiène, and final cost.
Material choice influences far more than strength. It changes weight, efficacité de l'isolation, nettoyabilité, durée de vie, and how the box behaves after repeated use. A rigid outer shell made from polyolefin resins can improve impact resistance and hygiene. Foam-based bodies can reduce mass and improve insulation, but the best answer depends on whether the shipment is one-way, consigné, or somewhere in between.
You should also separate shell material from insulation system. A buyer may focus on polyethylene, polypropylène, PPE, PSE, polyuréthane, or VIP because those words appear in quotes, but they do different jobs. One material may provide the hard exterior, another the thermal barrier, and a third the sealing or cushioning layer. Good suppliers explain the full build, not only the headline resin name.
For temperature-sensitive shipping, material selection is not only about thermal conductivity. It is also about water uptake, resistance to cleaning chemicals, stabilité dimensionnelle, récupération après impact, and how easy it is to maintain quality from lot to lot. That is why an apparently simple ice box deserves a more technical conversation than many buyers expect.
For buyers comparing samples, dimensions alone are not enough. Two boxes with similar external size can differ sharply in usable payload space because wall thickness, corner reinforcement, and lid design steal volume differently. If the route involves repeated reuse, impact resistance and closure durability matter as much as nominal insulation. If the route is one-way parcel distribution, cube efficiency and empty weight may matter more. This is why material language should always be tied back to the operating model, not treated as a marketing label.
How thermal performance should really be judged
A cold-chain box does not perform in isolation. The refrigerant, pack conditioning method, température de la charge utile, taux de remplissage, espace de tête, fréquence d'ouverture, and outside environment all change the result. That is why experienced buyers avoid broad claims like “keeps cold for 72 heures” unless the supplier can explain exactly under what loading and ambient assumptions that statement was measured.
Thermal performance claims should be tested as a system claim, not accepted as a box claim. In cold-chain development, le conteneur, the refrigerant, la charge utile, and the packout pattern interact. Industry development protocols such as ISTA 7D are used to compare how packaging performs under temperature cycles, but that kind of test is still only one step. Buyers need results that resemble the real route, charge utile réelle, and real handling pattern.
For vaccine and pharmaceutical moves, the performance conversation must go beyond hold-time marketing. Public guidance from WHO and CDC treats cold boxes and vaccine carriers as passive containers that rely on coolant packs, and CDC distinguishes qualified containers and packouts from ordinary food or beverage coolers. That distinction matters because a well-built shell can still fail if the coolant is wrong, the payload is warm, or the operator opens the box too often.
A serious supplier should be able to explain which variables were controlled during testing: température de départ du produit, pack conditioning method, profil ambiant, placement du capteur, lid-open events, et critères d'acceptation. Si la réponse est vague, the thermal claim is probably too generic to support a high-risk shipment.
Vaccine transport deserves extra caution because some liquid vaccines can be permanently damaged by freezing, even when a shipment still appears cold enough. WHO procurement guidance and CDC transport guidance both stress correct coolant preparation, planification d'itinéraire, et surveillance. The safest buying approach is to ask how the box is intended to be packed, what temperature range it is meant to support, and what evidence exists for that exact use.
In immunization work, cold boxes are passive devices rather than powered refrigerators. UNICEF procurement guidance describes cold boxes as insulated containers lined with coolant packs, with capacity and cold-life categories defined under controlled conditions. That is useful context for buyers: product class definitions exist, but real field performance still depends on route duration, exposition ambiante, and operator discipline.
If you are sourcing for vaccines, do not assume a rugged cooler is automatically appropriate. Ask whether the proposed configuration is meant for short transfer, off-site clinic use, emergency relocation, or last-mile outreach. Those scenarios can require different packout logic, monitoring frequency, et critères d'acceptation.
Documentation, cohérence, and why technical answers matter
In cold-chain buying, technical ambiguity usually turns into commercial risk later. If a supplier cannot say which drawing revision was approved, which material is locked, or how future changes are communicated, the buyer has very little protection when a later lot behaves differently. This matters even for ordinary reusable boxes, and it matters even more for medical or tightly specified programs.
The strongest suppliers do not only send a quotation. They explain assumptions. They identify what was tested, what was not tested, which dimensions are functional, which options are custom, and how they manage reorders. That level of clarity shortens internal approval cycles because procurement, opérations, and quality can review the same facts instead of arguing over marketing language.
Pour les acheteurs, la leçon est simple: treat documentation as part of performance. A container that is technically good but poorly documented may still be the wrong buy if your team depends on stable repeat supply, traçabilité, or future route qualification work.
Where this type of ice box fits well – and where it does not
A good sourcing decision starts by matching the box to the route, not by forcing one container into every job. Dans de nombreux cas, insulated ice boxs work best when you need passive temperature protection for a defined time window, physical durability, and a packout that operators can follow without guesswork.
Vaccine transport requires disciplined packout, surveillance de la température, et contrôle d'itinéraire. A rugged box alone is not enough. Buyers should distinguish a protective outer container from a qualified transport solution supported by the right coolant, instructions, et dispositifs de surveillance.
A useful way to compare options is to picture one real route instead of one abstract box. Par exemple, a same-day commercial loop with repeated door openings values durability, ergonomic handling, et lavabilité. A two-day one-way parcel route values insulation efficiency, dimensional optimization, et densité du fret. A vaccine transfer between facilities values packout discipline and temperature monitoring more than branding or cosmetic finish.
En pratique, the wrong box is often chosen because buyers compare catalog categories rather than shipping scenarios. The container that looks perfect for frozen seafood may not suit direct-to-patient medicines. The container that survives a warehouse shuttle may be a poor fit for outreach immunization where the operator carries the load by hand. Scenario-first sourcing reduces those mistakes.
Pour les vaccins, common use cases include planned transport to an off-site clinic, short emergency relocation, or controlled movement between facilities. Those are very different from parcel shipping, and they should not be supplied with ordinary picnic-style coolers or unmonitored improvised packouts.
What is shaping procurement decisions right now
Across temperature-sensitive logistics, buyers are putting more weight on route fit, répétabilité, and waste control than they did when a simple static hold-time claim was enough. That does not mean the market suddenly wants the most advanced packaging in every case. It means teams are trying to avoid false economy. They want fewer damaged shipments, fewer overbuilt packouts, better use of pallet space, and more predictable ordering.
That shift changes supplier evaluation. Vendors that can only repeat catalog language tend to struggle once buyers ask for usable volume under load, real shipping assumptions, or sample-to-production consistency. Suppliers that can talk clearly about geometry, refrigerant loading, surveillance, and operating constraints usually create more trust, even if their first quote is not the lowest.
Sustainability is part of the same conversation. A reusable box does not become sustainable just because it can technically survive multiple cycles. It becomes sustainable when return logistics, discipline de nettoyage, and replacement planning actually work. Many buyers now compare material choice and reuse strategy together instead of treating them as separate topics. That tends to produce more realistic packaging programs and more defensible purchasing decisions.
A practical supplier checklist before you place a bulk order
A good shortlist is built by asking every bidder for the same operating facts. That exposes weak offers quickly and makes commercial comparison much cleaner.
- Ask how drop resistance, load handling, or repeated-use durability are evaluated in routine QC.
- Separate ex-works unit price from delivered cost, coût des accessoires, and validation or documentation cost.
- Request cleaning and hygiene guidance, especially for food, Soins de santé, or multi-use return loops.
- Request sample-to-production control and change-control commitments, not only a first-sample promise.
- Clarify whether labels, codes à barres, RFID positions, molded recesses, and other traceability features are standard or custom.
- Ask which material belongs to the shell, which belongs to the insulation layer, and whether any substitutions are allowed after approval.
- For temperature-sensitive applications, ask for the recommended refrigerant type, méthode de préconditionnement, monitor placement, et hypothèses d'itinéraire.
- Verify lid style, gasket or closure method, drain design if relevant, and whether the unit remains stable when stacked fully loaded.
- Confirm internal dimensions, dimensions extérieures, and usable volume after coolant and inserts are loaded.
- Get a written answer on MOQ, délai de livraison de l'échantillon, délai de production, tooling cost if any, and what happens if forecasts move.
- Verify whether the seller controls production directly or is reselling a third-party design, and clarify who will answer technical issues after delivery.
Use the answers to normalize every quotation into the same comparison frame. Once each supplier is speaking to the same dimensions, same packout assumptions, same service level, and same delivery basis, price comparisons become much more meaningful.
First cost vs total cost of ownership
The visible invoice is only one layer of cost. With insulated ice boxs, total cost usually includes replacement rate, travail d'emballage, monitor use, densité du fret, empreinte de stockage, and sometimes reverse-logistics effort for returnable formats. A slightly heavier or more expensive box may still lower total cost if it reduces damage, improves stacking, or survives a larger number of loops.
This is particularly true when buyers compare reusable and semi-disposable options. Reuse counts on paper do not matter unless the container comes back in usable condition, can be cleaned without excessive labor, and re-enters the right route quickly. A low first-unit price can also become expensive if the design wastes pallet space, needs more coolant, or requires operators to improvise around weak closures or awkward loading.
Ask suppliers to help you model the packaging program in operational terms: unités par palette, empty return efficiency, recommended coolant quantity, sample failure rate, field breakage risk, and what documentation or reordering support exists after launch. That conversation often reveals more economic value than another round of price bargaining.
For higher-risk shipments, total cost must also account for excursion risk. One compromised medical shipment can erase the savings from many cheaper boxes. That is why buyers in these categories often pay attention to documentation quality, surveillance, and configuration discipline rather than choosing only on unit price.
FAQ
Can a vaccine ice box be treated like an ordinary cooler?
Non. Vaccine transport requires the right coolant preparation, dispositif de surveillance, and packout instructions. A rugged shell alone is not enough.
What should I ask before buying a vaccine box?
Ask for intended temperature band, durée de l'itinéraire, type de liquide de refroidissement, placement du capteur, and whether the configuration is qualified or only recommended.
Why is freezing risk mentioned so often?
Because some liquid vaccines can lose potency after exposure to freezing temperatures even if the product still appears cold.
Closing perspective
The best response to vaccine ice box vendor price is rarely a single advertised figure. Good buying decisions come from understanding the construction, the shipping logic, le vrai itinéraire, and the degree of control behind the quote. When those elements are clear, you can compare suppliers on something more meaningful than a headline price and choose a box that performs in real operations, not only in sales language.
À propos du tempk
We focus on cold chain packaging for food and temperature-sensitive shipments, including insulated shipping boxes, packs de glace, sacs plus frais, and related passive packaging components. Our work is most useful when buyers need a practical conversation about packout logic, insulation format, and route fit rather than a generic catalog answer. For projects that need custom sizing or a more application-specific configuration, we prefer to start from shipment requirements and operating reality.
Prochaine étape
If your team is narrowing suppliers, request one comparable packout assumption from each bidder before you compare prices.