Choosing Thermal Gel Pack Hospital Manufacturer for Real Cold-Chain and Handling Conditions
If you need a practical answer on thermal gel pack hospital manufacturer, start by separating what the pack can do on its own from what the full shipping or handling system still has to control.
For hospital procurement, patient-care hot/cold therapy, and internal healthcare handling, the most reliable answer is usually the same: choose the pack only after you know the target temperature band, the route duration or handling window, the geometry of the payload, and the level of documentation your team needs. That keeps sourcing tied to performance rather than to marketing language.
In a hospital setting, a thermal gel pack usually refers to a reusable hot/cold therapy pack used for pain relief, swelling control, post-operative recovery, or localized patient comfort. Some buyers also use similar packs for short internal transfers of temperature-sensitive items, but that is a different use case with different validation needs.
The first procurement question is therefore functional intent. Are you buying a patient-contact therapy pack, an instant emergency-use cold pack, or a refrigerant component for internal transport? The words sound similar, but the required materials, quality documentation, and performance expectations are not the same.
What it is – and what it is not
In hospitals, a thermal gel pack may look simple, but the intended use changes the documentation burden. A pack used for patient-contact hot or cold therapy sits in a different risk context from a pack used only to support transport, staging, or temporary cooling of supplies.
That is why hospital sourcing should not assume that every cold pack is interchangeable. The pack has to fit the care environment, cleaning routine, staff workflow, and labeling expectations. If it is marketed as a medical device, intended purpose and applicable conformity requirements may also matter.
Where the product is presented for patient-contact therapy or clinical use, documentation expectations may rise substantially. Intended purpose, labeling, and the relevant quality-system controls matter more than the visual similarity between one gel pack and another.
The main performance drivers buyers should understand
Hospital thermal packs sit at the intersection of material science and workflow design. The fill system has to store enough energy for useful therapy, but the gel also has to remain flexible enough for anatomy contact and safe enough for routine patient handling. That is why the same pack chemistry does not suit every healthcare use.
Thermal performance comes from more than the inner fill. The shell or film must stay flexible enough for the intended conditioning state, resist puncture or seam fatigue, and preserve a repeatable geometry around the payload. Even a good refrigerant chemistry can disappoint if the filled shape changes too much after freezing, if the cells distribute mass unevenly, or if the exterior becomes difficult to handle in the real workflow.
Geometry is especially important because heat does not enter a shipper or handling setup uniformly. Flat formats create broader contact and can reduce dead space. Thicker bricks or denser packs may store cooling energy longer, but they also occupy more volume and may create colder local contact points. The correct balance depends on whether you need even coverage, longer hold time, easier packing, cleaner handling, or a more controlled temperature window.
Outer construction matters just as much as the fill. The film or laminate must survive repeated bending at low temperature, resist pinholes, and accept cleaning. If the product includes a fabric sleeve or no-sweat layer, that feature may improve comfort or moisture control but can also change drying time and sanitation practice.
Matching the format to the use case
The best-fit use case depends on the trade-off you care about most: coverage, duration, handling, repeatability, condensation control, receiver experience, or tighter temperature buffering. The common patterns below help buyers compare those trade-offs quickly.
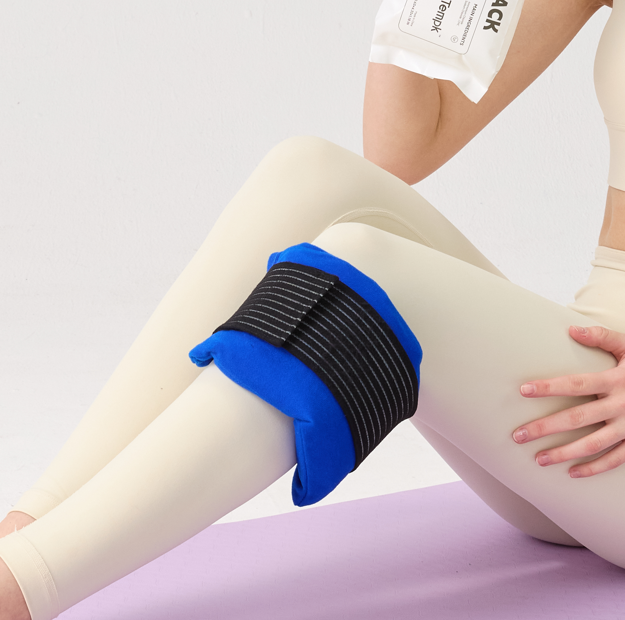
Patient care and post-operative recovery
Hospitals often want a pack that remains pliable when cold, distributes temperature evenly, and fits anatomical areas such as shoulders, knees, backs, or IV sites. In this context, comfort, surface finish, and ease of cleaning matter almost as much as how long the pack stays cold.
A pack that feels too rigid after freezing or develops very hard cold spots can create poor patient experience and extra nursing work. Shape, gel distribution, and sleeve compatibility therefore deserve attention during supplier qualification.
Central supply and ward handling
A hospital also needs repeatability. Packs may be frozen in one area, issued on another floor, wiped down after use, and returned for the next patient. That means the material must tolerate frequent handling and a realistic sanitation routine.
If the pack is single-patient use, packaging and labeling must make that clear. If it is intended for repeated use, staff need a practical inspection rule for leaks, seam damage, or surface degradation.
Healthcare logistics but not full cold-chain qualification
Some facilities ask whether the same thermal gel pack can also support internal medicine transfer or specimen handling. Sometimes it can help, but that does not make it a qualified pharmaceutical shipper. A therapy pack is a protective temperature aid, not an automatic substitute for a documented insulated shipping system.
When medicines, blood products, or laboratory specimens are involved, route, temperature range, and compliance rules still need separate evaluation.
A practical supplier checklist before scale-up
When hospital buyers, clinical procurement teams, and healthcare distributors buy in volume, the best supplier conversation is detailed and specific. It should cover dimensions in conditioned use, material choice, closure or seam quality, handling stress, lot traceability, and the practical instructions needed for the people who will freeze, pack, move, clean, or receive the product. A short list built on those points is usually more reliable than a long list built only on price and MOQ.
Most buying errors happen when teams compare packs before they have written down the real operating requirement. For hospital procurement, patient-care hot/cold therapy, and internal healthcare handling, the decision should start with whether the pack is intended for patient-contact therapy, transport support, or both, then move through pliability after conditioning and how evenly the cold or heat is distributed across the surface, cleanability, wipe-down compatibility, and whether the pack can tolerate the facility routine, and the handling realities behind lot traceability, labeling, and documentation expected by hospital procurement. If the shipment or use case has a visible end user, the evaluation should also include availability of multiple shapes and sizes that map to actual clinical protocols. That sequence keeps the discussion tied to route outcome rather than to catalog language.
Ask if the product is sold into regulated healthcare settings and what documentation accompanies each lot.
Request the material description for the outer film or textile interface and confirm whether it is latex-free or otherwise screened for common facility requirements.
Check how the pack behaves after freezing: pliable, semi-rigid, or rigid. This changes patient comfort and fit.
Confirm cleaning instructions and whether surface printing, sleeves, or seams tolerate repeated wipe-downs.
Verify carton labeling, unit-of-measure consistency, and traceability support for hospital inventory systems.
If the product is marketed as a medical device in your target market, ask for the applicable registration, declaration, or conformity documents.
Run a ward-level user trial before full rollout. Nursing teams often identify handling issues that are invisible in a purchasing meeting.
For long-term procurement, change control matters almost as much as first-pass performance. Buyers should know what happens if the supplier changes film structure, resin grade, fill formulation, seal pattern, artwork, or production site. If those changes are not communicated and re-evaluated, a successful pilot can drift into a less reliable production result without anyone noticing until the field complaints begin.
Qualification and documentation points that matter
ISO 13485 is widely used as the quality management benchmark for the design and manufacture of medical devices. If a thermal gel pack is marketed as a medical device or sold into tightly controlled healthcare channels, buyers often look for this type of quality-system discipline because it supports traceability, risk control, and consistent production.
In Europe, if the product is actually placed on the market as a medical device, buyers should also expect the appropriate CE-marking documentation for that intended use. The important caveat is intended purpose: not every cold pack is a medical device simply because it is used in healthcare.
For internal hospital transport of medicines or samples, remember that a gel pack is still only one component. A therapy pack alone does not satisfy the same requirements as a documented insulated or qualified transport system.
A useful supplier data pack normally includes conditioned dimensions, nominal fill weight or range, material description, conditioning instructions, recommended use window, storage guidance, and any relevant test information on leakage, durability, or route performance. For regulated or quality-sensitive programs, buyers often also want lot traceability, revision control on specifications and artwork, and a clear statement of what the supplier has validated versus what still needs route-specific qualification by the shipper.
Qualification should mirror the lane you actually plan to run. That means defining the payload temperature at packout, the number and location of refrigerants, the insulation configuration, the expected transit duration, and the most credible exposure profile. Temperature loggers or other monitoring tools help confirm whether the packout protects the target range at the warmest and most vulnerable locations, not only at the geometric center of the shipper.
Hospital teams often benefit from testing that goes beyond nominal hot or cold duration. Useful checks include pliability after conditioning, surface temperature feel, wipe-down tolerance, seam reliability, and whether the pack keeps its shape after repeated use in carts, wards, and treatment rooms.
For procurement, sample review should involve the real clinical routine. A pack that is comfortable in a desktop demonstration may become impractical if it is too stiff after freezing, too heavy for routine bedside handling, or too fragile for repeated transport between departments.
Common questions
What makes a thermal gel pack suitable for hospital use?
The right balance of thermal performance, patient comfort, cleanability, traceability, and documentation. Hospital suitability is about workflow control, not only how cold the pack gets.
Does hospital use automatically mean the pack is a medical device?
No. The regulatory status depends on the intended purpose and how the product is placed on the market. Buyers should ask the supplier to define that status clearly.
Can one hospital gel pack cover both therapy and shipping needs?
Sometimes it can support both roles, but they should not be treated as identical applications. Transport of medicines or specimens usually requires separate packaging evaluation and documentation.
Final word
For hospital procurement, the right thermal gel pack is the one that fits the clinical protocol, survives the sanitation routine, and comes with clear documentation. In healthcare, usability and traceability matter as much as temperature retention.
The strongest procurement outcome usually comes from matching the refrigerant to the exact route or use case, then testing the result under realistic conditions, and finally choosing the supplier that can reproduce that result consistently. That approach is slower than buying by catalog description, but it is usually much cheaper than troubleshooting failures after launch.
About Tempk
At Tempk, we work across temperature-controlled packaging and personal thermotherapy-related product categories. Our website presents gel pack and PCM-based solutions alongside insulated shipping products and custom development resources. That mix is helpful when a healthcare buyer needs a supplier who understands both practical pack design and the broader temperature-management workflow around it.
Next step
If you are reviewing suppliers, ask for a proposal that matches the intended use, conditioning routine, packaging format, and documentation level you actually need.































